 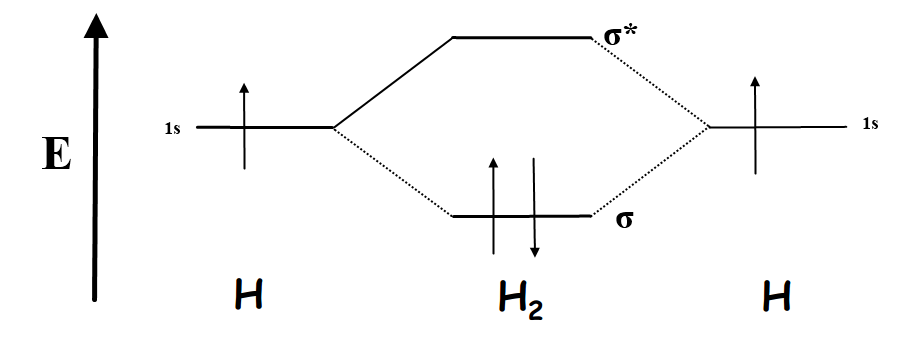
Each 2p orbital is oriented along a single axis. Each of the three 2p atomic orbitals in each nitrogen atom must overlap to form a bonding molecular orbital, to accommodate three electron pairs. Now consider the molecular orbital description of bonding in \(N_2\). The result, from valence shell electron pair sharing concepts, is that three pairs of electrons are shared between two nitrogen atoms in a “triple bond.” Intuition suggests that the triple bond in \(N_2\) should be much stronger than the single bond in H 2 or in \(F_2\). Each of these unpaired electrons is available for sharing with a second nitrogen atom. A nitrogen atom has three unpaired electrons in its valence shell the three 2p electrons distribute themselves over the three 2p orbitals, each oriented along a different axis. The unusually strong bond in nitrogen can be explained using both the valence shell electron pair sharing model and electron orbital descriptions. The bond energy of \(N_2\) is 956 kJ mol -1 this is much larger than the bond energy of H 2, 458 kJ mol -1, and of \(F_2\), which is 160 kJ mol -1. Recall that the bond energy (or bond strength) is the energy required to separate the bonded atoms. If the electron being ionized has a higher energy in \(F_2\) than in F, why is \(F_2\) a stable molecule? A more complete description of the molecular orbital concept of chemical bonding is required.īond energies are next considered. This seems inconsistent with the bonding orbital concept developed above, which states that the electrons in the bond have a lower energy than in the separated atoms. In this case, find that the ionization energy of molecular fluorine is 1515 kJ mol -1, which is smaller than the ionization energy of a fluorine atom, 1681 kJ mol -1. Once again, the energy of the electrons in molecular nitrogen is lower than that of the electrons in the separated atoms, so the molecule is bound.Īs a third example, consider fluorine, \(F_2\). The ionization energy of molecular nitrogen is 1503 kJ mol -1, and that of atomic nitrogen is 1402 kJ mol -1. This conclusion is consistent with the view of shared electrons in bonding molecular orbitals.Īs a second example, consider the nitrogen molecule, \(N_2\). Hence, energy is required to break the bond, and the molecule is bound.Ī bond is formed when the energy of the electrons in the molecule is lower than the energy of the electrons in the separated atoms. To pull the atoms apart, the energy of the electron must be increased. Therefore, it requires more energy to remove an electron from the hydrogen molecule than from the hydrogen atom the electron therefore has a lower energy in the molecule. This number is primarily important in comparison to the ionization energy of a hydrogen atom, which is 1312 kJ mol -1. The measured ionization energy of H 2 is 1488 kJ mol -1. This is the energy required to remove an electron, in this case, from a molecule: The energies of electrons in molecular orbitals can be observed directly by measuring the ionization energy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
